12 November 2015
 New interdisciplinary research has revealed the frontline role tiny algae could play in the battle against cancer, through the innovative use of nanotechnology.
New interdisciplinary research has revealed the frontline role tiny algae could play in the battle against cancer, through the innovative use of nanotechnology.
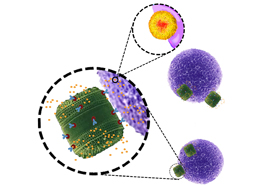
The team of Professor Nico Voelcker at the University of South Australia and collaborators in Dresden, Germany, have genetically engineered diatom algae to become therapeutic nanoporous particles, which, when loaded with chemotherapeutic drugs can be used to destroy cancer cells in the human body, without harming healthy cells.
Details of this research: "Targeted drug delivery using genetically engineered diatom biosilica” are being published in the latest edition of Nature Communications, this week and Professor Voelcker explains the potential benefits of this method to combat cancer.
“By genetically engineering diatom algae - tiny, unicellular, photosynthesizing algae with a skeleton made of nanoporous silica, we are able to produce an antibody-binding protein on the surface of their shells,” Prof Voelcker says.
“Anti-cancer chemotherapeutic drugs are often toxic to normal tissues. To minimise the off-target toxicity, the drugs can be hidden inside the antibody-coated nanoparticles.
“The antibody binds only to molecules found on cancer cells, thus delivering the toxic drug specifically to the target cells.”
As the Strand Leader in Biomaterials Engineering & Nanomedicine at the University of South Australia’s Future Industries Institute, Professor Voelcker highlights how this type of research could influence future health care:
“Although it is still early days, this novel drug delivery system based on a biotechnologically tailored, renewable material holds a lot of potential for the therapy of solid tumours including currently untreatable brain tumours.”
Media contact: Will Venn office (08) 8302 0096 email will.venn@unisa.edu.au
Nico Voelcker office (08) 8302 5508 email Nico.Voelcker@unisa.edu.au




